A private clinic in Chicago's Lincoln Park neighborhood is now offering coronavirus antibody tests, which the FDA says can help identify who has been infected as well as identify those still at risk.
Innovative Express Care believes it is one of the first clinics in the Chicago area to offer an FDA-approved test to the general public.
Antibodies may provide potential immunity against future infection, and the FDA says results from these tests can also help inform who may qualify to donate plasma to patients seriously ill with COVID-19.
Dr. Rahul Khare, an emergency room physician and the CEO of Innovative Express Care, said the information will be invaluable.
"I think it is very, very important to understand the prevalence of COVID-19 and get people back to work, especially healthcare workers and first responders," Dr. Khare said. "If you do have antibodies, what does that tell you? For right now, it [says] you have a low chance of getting [coronavirus] again."
The FDA has expanded access to Serology (antibody) tests through the emergency use authorization (EAU) process. Innovative Express Care is using a test called Euroimmun, from parent company PerkinElmer, which got its EAU on March 31.
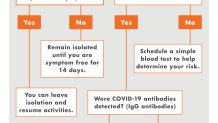
To qualify for an antibody test, patients cannot have had symptoms for at least two weeks.
"If you do have symptoms, we don’t want you to have the antibody test. Actually, it would give you a false sense of reassurance," said Dr. Khare. "The antibodies will not show up for at least 10-14 days."
Innovative Express Care has partnered with Quest Diagnostics to run its lab results. In a press release, Quest said it, "expects to scale up testing over the coming weeks. Laboratory capacity is expected to expand from approximately 70,000 tests per day by the end of this week to approximately 150,000 tests a day by early-May. Quest expects to report antibody test results within 1-2 days from specimen collection, depending on demand."
Local
Dr. Khare hopes to increase daily testing at his facility from 150 currently to as many as 300 in the coming weeks. He said more tests will give officials a better understanding of the prevalence of coronavrius and at achieving what is known as "herd immunity."
"We want 60 to 70 percent of people to have had antibodies to this. That we know for a fact there’s a significant decrease in contagiousness of this," he said.



